**The AGA grants EECG Research Awards each year to graduate students and post-doctoral researchers who are at a critical point in their research, where additional funds would allow them to conclude their research project and prepare it for publication. EECG awardees also get the opportunity to hone their science communication and write posts over their grant tenure for the AGA Blog. In the wrap-up to the series, awardees talk about their award and research in their ‘epilogue’.**
 About the Blog Author: Jessi Rick is a PhD candidate in Dr. Katie Wagner’s lab and the University of Wyoming’s Program in Ecology. She is fascinated by how environmental change drives both micro- and macroevolution. She completed a BS in Organismal Biology at the University of Arizona and an MS in Integrated Biosciences from the University of Minnesota- Duluth. She has researched the population genetics of organisms from crop pests to gray wolves to fishes. She loves learning how to uncover the secrets hidden in genomes. You can learn more about Jessi’s work on her website and find her tweeting at @jessi_rick.
About the Blog Author: Jessi Rick is a PhD candidate in Dr. Katie Wagner’s lab and the University of Wyoming’s Program in Ecology. She is fascinated by how environmental change drives both micro- and macroevolution. She completed a BS in Organismal Biology at the University of Arizona and an MS in Integrated Biosciences from the University of Minnesota- Duluth. She has researched the population genetics of organisms from crop pests to gray wolves to fishes. She loves learning how to uncover the secrets hidden in genomes. You can learn more about Jessi’s work on her website and find her tweeting at @jessi_rick.
East Africa’s Lake Tanganyika is the deepest, oldest, and volumetrically largest of the African Great Lakes. It is well-known by evolutionary biologists for its incredible diversity of cichlid fishes, which have diversified into species with a wide array of colors, feeding strategies, and habitat affinities. However, Lake Tanganyika is also home to numerous other species flocks that have evolved within the lake, including a four-species radiation of open-water predatory fishes in the genus Lates. These piscivores live in deep-water environments throughout the lake, have been recorded at sizes up to two meters long, and are important components of Lake Tanganyika’s fishery, the second largest inland fishery on the African continent.
I was honored to receive an AGA EECG award to work toward understanding the roles that climate and hydrogeological change have played in the evolutionary history of the Lates clade in Lake Tanganyika. I am excited to have the first part of that work—focused on understanding the genomic population structure of these species—coming out soon in Journal of Heredity. In our paper, we find that three of the species (L. angustifrons, L. microlepis, and L. stappersii) show little geographic population structure, while the fourth species L. mariae appears to have a genetically distinct population that occurs in the southern reaches of the lake. In addition, we find weakly differentiated groups that occur sympatrically in all four species. These results have potentially important implications for understanding the conservation and management of these four fish species, as well as for understanding the role that historical and contemporary environmental conditions play in shaping species’ population genomic patterns.

Although my dissertation research focuses on a single body of water, sampling fish from such a large lake is challenging. Most previous sampling effort from our group and collaborators has focused on the area around Kigoma, Tanzania. However, sampling only a small stretch of the lake would not allow characterization of the lake-wide population structure of the wide-ranging Lates fishes across the whole 673km length of the lake. In 2018, I therefore took advantage of an opportunity to join a research cruise organized by Swiss collaborators from EAWAG (the Swiss Federal Institute of Aquatic Science and Technology) that traveled the entire length of the Tanzanian shore to sample water and plankton, as well as collect fish and mollusks for genetic and stable isotope analyses.
Traveling for two weeks aboard a cargo-turned-research vessel—with an international team of researchers from Tanzania, Switzerland, Germany, and Canada with a Congolese crew—was an amazing experience. I learned an incredible amount about my study species from the Tanzanian researchers from the Tanzanian Fisheries Research Institute (TAFIRI) working on the lake, as well as our interactions with local fishermen in villages along the way. I saw juvenile Lates in their nursery habitats while snorkeling near the shore and observed firsthand the detrimental effects of beach seining on juvenile fish populations. From sampling fish and water in parallel, I also saw how the lake physically changes as we moved from north to south, with changes in water column stratification, oxygenation, and primary productivity (Ehrenfels et al. 2021), as well as how the relative abundance of each of the Lates and sardine species changed as we made our way southward.
From the genetic data, it is clear that fish from all four species move throughout the lake with ease. There is no pattern of isolation-by-distance (which would suggest restricted movement or gene flow), which is good news for the Lake Tanganyika fishery. We did find, however, that L. mariae that were collected at our southernmost sampling site are genetically differentiated from L. mariae in the rest of the lake. While we don’t have definitive answers as to what may have caused this genetic differentiation, biogeochemical differences between the southern basin and rest of the lake warrant further exploration. In particular, we lack good records of the timing and location of spawning for the four species, which would be highly valuable for testing hypotheses related to the causes of intraspecific differentiation and should be a focus of future monitoring and genetic work.
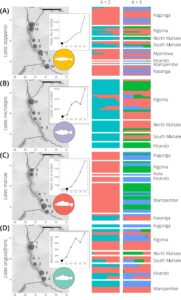
Another interesting piece to the puzzle is that Lake Tanganyika is a rift lake that began forming ~9-12 million years ago (Cohen et al. 1993) and has been dramatically affected by global climatic changes over the course of its long history. In the past, glacial cycles have corresponded to megadrought conditions in sub-Saharan Africa, and likely lowered the water levels in Lake Tanganyika hundreds of meters (Burnett et al. 2011, McGlue et al. 2008). The lake has three major basins, which are delineated by underwater ridges and which would have separated the lake hydrologically and physically—and in turn, altered connectivity between populations of fishes in different parts of the lake—during extreme drought periods. While the population genomic structure of the four species suggests that these fish have not retained a geographically consistent signal of the three-basin separation, it is possible that the weak sympatric population structure that we see in all four species may be a result of this history. This will be another fascinating hypothesis to test in future work.
As with most studies, even though we understand a little bit more about these fishes from their genomics, our results left us with many further hypotheses to investigate in this system. Thus far, we’ve learned that these large predators impact their ecosystem—including the human populations surrounding the lake—and are impacted by it, and I’m excited to dig in more to disentangling these influences as I work on completing my PhD research.
References



