Among vertebrate animals, sexual reproduction is ubiquitous.
But why? Asexual populations should outcompete their sexual neighbors (Maynard Smith, 1958).
Over the past century, scientists batted around ideas explaining the scarcity of asexual reproduction, which led to numerous hypotheses regarding the benefits of sex (here is a non-exhaustive list). Several empirical studies examining the cost of asexuality in vertebrates with respect to endurance between closely related asexual and sexual species indicate that asexuals perform worse in aerobic activities (e.g., mole salamanders [Denton et al., 2017], dace [Mee et al., 2011], and whiptail lizards [Cullum, 1997]; Fig. 1).

This prompts the question– Is a common biological underpinning responsible for decreased endurance capacity in asexual species?
To examine this question, you might ask yourself– What fine-scale process might be powering differences in aerobic physiological performance between sexual and asexual organisms? Reaching back into your high school biology memories, you might say: Power… powerhouse… the powerhouse of the cell! Mitochondria! Alright, maybe it didn’t happen exactly like that– but if you’re like me you might find yourself wondering if mitochondria might be associated with the observed performance differences in asexual vs sexual organisms.
So what is the relationship between mitochondria and aerobic activity (such as endurance)?
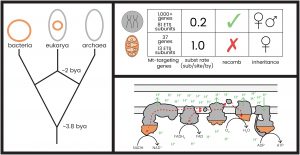
Mitochondria: Two billion years ago an archaeon engulfed an alphaproteobacterium; a paramount evolutionary event at the base of all known eukaryotic organisms (Fig. 2, left). Because of this event, most eukaryotes have two genomes with unique evolutionary histories: nuclear (archaeal origin) and mitochondrial (alphaproteobacterial origin). Mitochondrial respiration, the process by which eukaryotes generate cellular energy in the form of ATP, depends on the compatibility of gene products from these two genomes (mitochondrial: 37 genes; nuclear: 1,000+ genes). Co-evolved gene products from the mitochondrial and nuclear genomes interact to facilitate electron flow and energy production, thus providing power for virtually all cellular (and thus organismal) activities (e.g., electron transport chain in Fig. 2, bottom right).
Endurance:Because ATP production at the electron transport system is eighteen times greater than that of glycolysis, mitochondria provide the vast majority of energy for biological work in eukaryotes. The co-evolution of mitochondrial and nuclear gene products is therefore crucial for intense aerobic activity such as endurance performance, which is evident in studies relating mitochondrial function and endurance (Gollnick and Saltin, 1982; Eynon et al., 2011).
Okay, but what about sex?
Sex: Because the mitochondrial genome acquires deleterious mutations at a higher rate than the nuclear genome (due to a higher mutation rate, lower effective population size, and lack of genetic recombination), selection in sexual eukaryotes favors chromosomal regions with compensatory nuclear mutations via genetic recombination (Felsenstein, 1974; Kimura, 1963; Lynch and Gabriel, 1990; Marais, 2007; Fig. 2 upper right). Asexual lineages lack the ability to efficiently filter compensatory nuclear alleles via genetic recombination, and thus are predicted to gradually lose compatibility between mitochondrial and nuclear genomes (Havird et al., 2015).
The universal need for energy among vertebrates suggests that this intragenomic network may underpin the low frequency of obligately asexual organisms.

Whiptail lizards (genus Aspidoscelis; Fig. 3 left) are a great study system for examining the costs of asexuality in vertebrates. Squamate reptiles (snakes, lizards, & amphisbaenians) are the only vertebrate group with species that reproduce parthenogenetically (asexual reproduction with absolutely no male input), and one third of all whiptail lizards, a genus of ~40 species, reproduce asexually. These unique lizards are distributed throughout the deserts of North America, which vast, beautiful landscape is the setting for my field work (Fig. 3 right).
I am examining the associations of molecular, physiological, and organismal traits involved in the decline of aerobic performance in asexual vertebrates, specifically in the context of oxidative phosphorylation facilitated by mitochondrial respiration. My preliminary findings of endurance in whiptail lizards corroborate those of previous studies (i.e., lower endurance capacity in asexuals), and also indicate reduced mitochondrial efficiency in asexuals. The AGA-EECG award is supporting the functional genomic aspect of my research, wherein I will (A) examine differential gene expression of asexual whiptails in the context of an endurance experiment and (B) test for differences in selection at mitochondrial-targeted genes. I’ll leave a detailed telling of this experiment and my findings for my next blog post – stay tuned!
References:
Felsenstein J. The evolutionary advantage of recombination. Genetics. 1974;78(2):737-756.
Maynard Smith J. The Theory of Evolution. Cambridge University Press; 1958.

Randy is a Ph.D. candidate at Auburn University working in the labs of Tonia Schwartz and Jamie Oaks. He is broadly interested in understanding the agents responsible for earth’s biodiversity and the evolutionary path to a phenotype (genomic, transcriptomic, physiological, and organismal), and uses techniques in genomics, phylogenetics, and physiology to answer questions within this realm of biology. He was awarded the EECG in 2020 for his work on examining molecular, physiological, and organismal traits involved in decline of performance in asexual whiptail lizards.




