**The AGA grants EECG Research Awards each year to graduate students and post-doctoral researchers who are at a critical point in their research, where additional funds would allow them to conclude their research project and prepare it for publication. EECG awardees also get the opportunity to hone their science communication and write posts over their grant tenure for the AGA Blog. In the first in the series, our EECG awardees write about their research and their interests as an ’embarkation’.**

About the Blog Author: Omid is a PhD student in the Brisson lab at the University of Rochester. He is too broadly interested in how evolutionary processes generate and maintain variation from populations into species in natural populations, and how development relays genetic variation into phenotypic variation in changing environments. Worst yet, his twitter handle is @evodevomid.
One of the big questions at the interface of evolutionary and developmental biology (‘evo-devo’) asks to what extent adaptations first arise from environmentally induced phenotypic variation (see Laland et al., 2014, Laland et al., 2015, Pfennig, 2021, and Schwander and Leimar, 2011). This type of plastic variation, called developmental plasticity, can persist through generations with no changing genetic component so long as the environmental pressures also persist. As genetic variants arise in a population which favorably contribute to plastic variation, adaptive evolution that involves gene-frequency changes follow. This process of genetic variation spreading in a population to bolster plastic variation is called genetic accommodation (West-Eberhard, 2005).
We can’t begin to address how often plasticity actually precedes adaptations because we know too little about the evolutionary genetics of genetic accommodation itself, especially in natural populations. The difficulty here is an empirical one: studying accommodation requires one to either observe new adaptations arise or infer the processes that produced existing adaptations. For the former, new adaptations are too rare to wait around for in nature; while the challenge for the latter is that for a given adaptation, evolution by genetic change can’t always be disentangled from evolution by environmental persistence.
To circumvent these challenges, we can take an indirect approach by comparing existing adaptations that differ by being either under genetic or environmental control. As we compare increasingly similar adaptations, we increasingly reduce confounding differences. For example, while the same adaptive trait may be under genetic control in one species, and environmentally controlled in a closely related species, which differences can we ascribe to the process of accommodation directly?
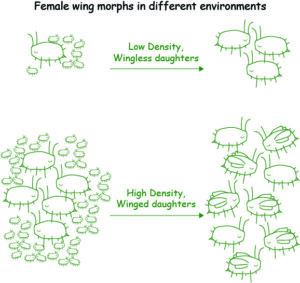
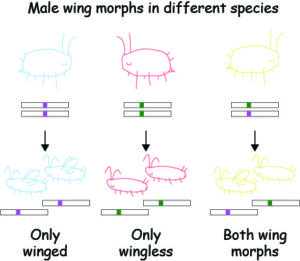
In my PhD work, I sought to use aphids to simplify this comparison considerably by studying their wing dimorphisms. The wing dimorphism refers to the production of either winged or wingless morphs in response to different cues. In females, this cue is the density of aphids on a host plant (see Figure 1). At high density, mothers start to produce wing morphs that migrate to new host plants. At low density, mothers produce a more fecund wingless morph. In males, the cue is genetic: the specific wing allele they carry determines the wing morph (see Figure 2). While all aphid species have some form of the environmentally controlled female wing dimorphism, the genetic male dimorphism is rare in comparison (about 4% of ~5,000 species, meaning most species produce only one male wing morph).
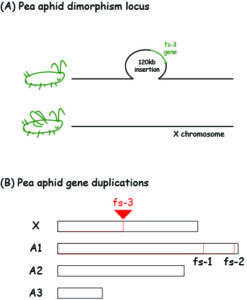
Our lab recently mapped the male wing dimorphism allele in pea aphids (Acyrthosiphon pisum, which have dimorphic males and females) to a 120 kb insertion, specific to wingless males (Li et al., 2020). The only gene in this (big) insertion is a gene called follistatin, and pea aphids have two other duplicates elsewhere in the genome (see Figure 3). You won’t be surprised that embryos homozygous for that wingless allele develop only into wingless males, and that embryos homozygous for the winged allele (no insertion present) develop only into wingless males. You may also not be surprised that female embryos (winged or wingless) don’t express this wingless male-specific allele with a follistatin duplicate. But imagine our surprise when we found that another duplicate of the follistatin gene (on a separate chromosome) has a similar expression profile between winged and wingless females (only expressed in wingless females reared a low density environments). So we have three follistatin gene duplications in pea aphids, one which serves the canonical essential function of follistatin, while the other two have specific roles in the environmental and genetic control of wing dimorphism in females and males, respectively.
And this is where the AGA EECG award comes into play. While we have a link between the genetic (male polymorphism) and environmental control (female plasticity) of the same trait (wing dimorphisms) in the same species (pea aphids) in the same genotype (males have only one X chromosome), what we want is to address which came first. Did the follistatin role in the female plasticity arise first and give rise to the genetic male dimorphism through gene duplication? Or did the male role of follistatin precede its role in the female plasticity? Stay tuned for my Epilogue post where (hopefully) I’ll report back about how I use comparative genomics in closely related species to study which gene duplicate began its role first in the wing dimorphism.
References
Laland, Kevin et al. 2014. “Does Evolutionary Theory Need a Rethink?” Nature 514(7521): 161–64.
Pfennig, David W. 2021. Phenotypic Plasticity and Evolution. Causes, Consequences, Controversies.



