
About the author
Dr. Jae Young Choi was a postdoctoral researcher in the lab of Dr. Michael Puruggan at New York University. His research focuses on understanding the evolutionary basis of island adaptive radiation in plants using genomics and fieldwork based approaches. In collaboration with Dr. Elizabeth Stacy at University of Nevada Las Vegas he is studying the Hawaiian Metrosideros as a model for understanding the biology underlying adaptive radiation. He has recently started a new role as an Assistant Professor at the University of Kansas. You can follow Jae on twitter @Choi_JaeY or visit his lab website.
Recombination is an important but understudied force of evolution
Meiotic recombination is the exchange of genetic material between the double-strand breaks of homologous chromosomes. Recombination shuffles the parental genotypes and generates novel allelic combinations that can be involved in various evolutionary scenarios (Dapper and Payseur 2017). Elucidating the effects arising from recombination is necessary for a deeper mechanistic understanding of evolutionary biology. A rich theoretical literature has predicted the consequences arising from recombination rate variation (Otto and Barton 1997; Otto and Michalakis 1998), but empirical studies have lagged. Currently, we have an incomplete knowledge on how recombination rate influences evolution in natural populations.
An evolutionary process where recombination is likely to play an important role is during adaptive radiations, where a common ancestor has undergone diversification into multiple adaptive forms (Schluter 2000). Organisms undergoing an adaptive radiation are ideal systems to study the mechanisms of adaptation and speciation. Recombination is predicted to play a dynamic role during the different stages of the radiation. In initial stages of adaptive radiation, elevated levels of recombination would favor the shuffling of ancestral variation and generate the diversity of adaptive forms, but in later stages suppression of recombination would be favored to couple the adaptive alleles (Berner and Salzburger 2015). But these are theoretical predictions and few empirical studies have investigated recombination rate variation and its evolutionary role during adaptive radiation.
Metrosideros is a model for studying the genomics of adaptive radiation.
Metrosideros is a woody species complex that dominates the mesic and wet forests of the Hawaiian Islands (Percy et al. 2008; Dupuis et al. 2019). From an evolutionary perspective, Hawaiian Metrosideros is a fascinating system to study because it exhibits the classical patterns of adaptive radiation across the archipelago, displaying striking phenotypic variation while inhabiting an extreme range of environments (e.g. cooled lava flow, high altitude, riparian zones, deserts). Across the Hawaiian Islands, there are more than 20 vegetatively distinct Metrosideros taxa or morphotypes (Dawson and Stemmermann 1990; Stacy and Sakishima 2019). The Metrosideros radiation is particularly interesting because despite being long-lived perennials, they have adapted rapidly to novel environments that became available as new volcanic islands formed (Stacy et al. 2014; Stacy and Sakishima 2019).
Recently, we have published a study that investigated the genomic basis of the Hawaii Metrosideros adaptive radiation (Choi et al. 2021). The evolutionary history of Hawaiian Metrosideros showed evidence of extensive reticulation associated with significant sharing of ancestral variation between taxa and secondarily with admixture. We investigated the genomic architecture underlying the adaptive radiation and discovered that divergent selection drove the formation of differentiation outliers in paired taxa representing early stages of speciation/divergence. Analysis of the evolutionary origins of the outlier SNPs showed enrichment for ancestral variations under divergent selection. Our findings suggest that Hawaiian Metrosideros possesses an unexpectedly rich pool of ancestral genetic variation, and the reassortment of these variations has fueled the adaptive radiation.
The missing piece: Recombination and its role during adaptive radiation.
Identifying genomic regions with increased genetic differentiation between incipient populations is critical for understanding the Metrosideros adaptive radiation and speciation history, because they will include the genes responsible for speciation and consequently form the molecular basis of adaptive radiation. For incipient species, especially those that are able to hybridize, recombination can be a strong factor determining the ancestry of the hybrid genome (Schumer et al. 2018). Because lineage sorting is faster in regions of low recombination, a reduction in recombination levels can result in increased levels of genetic differentiation between populations (Nachman and Payseur 2012; Cruickshank and Hahn 2014). Variation in recombination rate will complicate speciation research by creating genomic regions with increased divergence that are not associated with the genes involved in speciation. Hence, an important missing piece of my research has been the recombination landscape within the Metrosideros group, and teasing apart the role of selection versus recombination in shaping the speciation process.

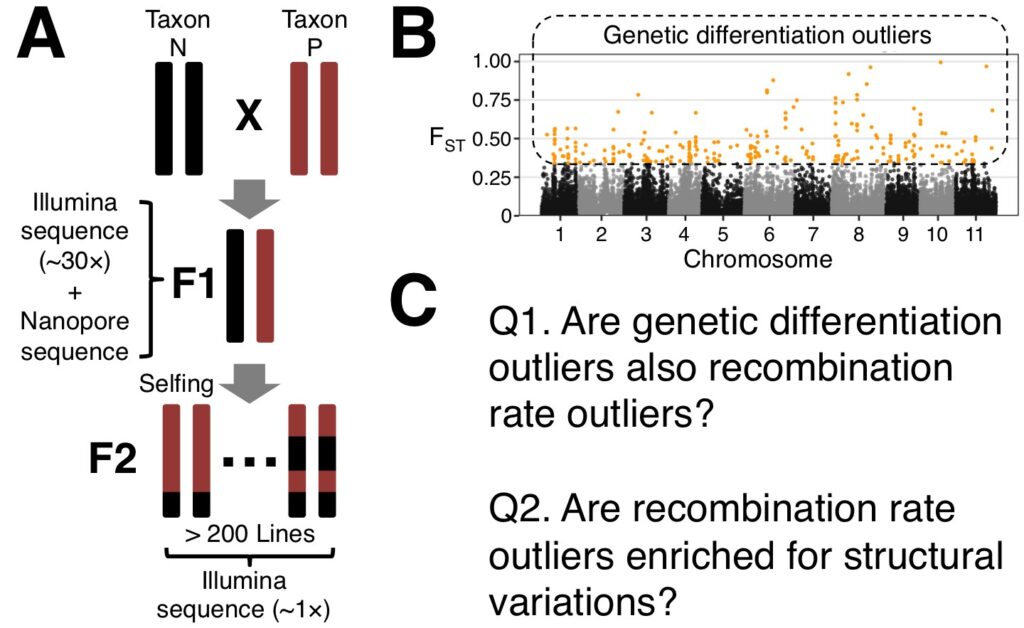
The EECG research award will support a project that will generate a fine-scale genetic map of the Hawaiian Metrosideros group (see Figure 1 for overall research project). This will be accomplished by genome sequencing F2 recombinants that were generated from two different parental crosses (Figure 1A). Two F1 progeny were selected for selfing and generate more than 200 F2 progenies (see Figure 2 for example individuals). The F1 and F2s will be whole genome sequenced and a Hidden Markov Model framework (Rowan et al. 2015) will be used to determine the haplotype and crossover events. The recombination map will be used to study how recombination has affected the genomic architecture underlying adaptive radiation (Figure 1B). Specifically we will be asking two major questions (Figure 1C):
Question 1: Do genetic differentiation outliers have significantly different recombination rate?
Question 2: Are genomic regions with outlier recombination rate enriched for structural variation?
References
Berner D, Salzburger W. 2015. The genomics of organismal diversification illuminated by adaptive radiations. Trends Genet. 31:491–499.
Choi JY, Dai X, Alam O, Peng JZ, Rughani P, Hickey S, Harrington E, Juul S, Ayroles JF, Purugganan MD, et al. 2021. Ancestral polymorphisms shape the adaptive radiation of Metrosideros across the Hawaiian Islands. Proc Natl Acad Sci U S A 118:e2023801118.
Cruickshank TE, Hahn MW. 2014. Reanalysis suggests that genomic islands of speciation are due to reduced diversity, not reduced gene flow. Molecular Ecology 23:3133–3157.
Dapper AL, Payseur BA. 2017. Connecting theory and data to understand recombination rate evolution. Philos Trans R Soc Lond B Biol Sci 372:20160469.
Dawson J, Stemmermann L. 1990. Metrosideros (Gaud). In: Wagner W, Herbst D, Sohmer S, editors. Manual of the flowering plants of Hawai’i. Honolulu, Hawai’i: Univ. Hawai’i Press. p. 964–970.
Dupuis JR, Pillon Y, Sakishima T, Gemmill CEC, Chamala S, Barbazuk WB, Geib SM, Stacy EA. 2019. Targeted amplicon sequencing of 40 nuclear genes supports a single introduction and rapid radiation of Hawaiian Metrosideros (Myrtaceae). Plant Syst Evol [Internet]. Available from: https://doi.org/10.1007/s00606-019-01615-0
Nachman MW, Payseur BA. 2012. Recombination rate variation and speciation: theoretical predictions and empirical results from rabbits and mice. Philos Trans R Soc Lond B Biol Sci 367:409–421.
Otto SP, Barton NH. 1997. The Evolution of Recombination: Removing the Limits to Natural Selection. Genetics 147:879–906.
Otto SP, Michalakis Y. 1998. The evolution of recombination in changing environments. Trends in Ecology & Evolution 13:145–151.
Percy DM, Garver AM, Wagner WL, James HF, Cunningham CW, Miller SE, Fleischer RC. 2008. Progressive island colonization and ancient origin of Hawaiian Metrosideros (Myrtaceae). Proceedings. Biological sciences 275:1479–1490.
Rowan BA, Patel V, Weigel D, Schneeberger K. 2015. Rapid and inexpensive whole-genome genotyping-by-sequencing for crossover localization and fine-scale genetic mapping. G3 (Bethesda) 5:385–398.
Schluter D. 2000. The Ecology of Adaptive Radiation. Oxford University Press
Schumer M, Xu C, Powell DL, Durvasula A, Skov L, Holland C, Blazier JC, Sankararaman S, Andolfatto P, Rosenthal GG, et al. 2018. Natural selection interacts with recombination to shape the evolution of hybrid genomes. Science 360:656–660.
Stacy EA, Johansen JB, Sakishima T, Price DK, Pillon Y. 2014. Incipient radiation within the dominant Hawaiian tree Metrosideros polymorpha. Heredity 113:334–342.
Stacy EA, Sakishima T. 2019. Phylogeography of the highly dispersible landscape-dominant woody species complex, Metrosideros, in Hawaii. Journal of Biogeography 46:2215–2231.



