**This post is a part of the series on the 2019 AGA Presidential Symposium – Sex and Asex: the genetics of complex life cycles**

About the author: Frank Stearns is an Adjunct Professor teaching Genetics and a writing course on Science Communication. He is interested in adaptation and speciation genetics and in the history of biology. He recently finished a postdoc at Johns Hopkins University. He has written for the AGA Blog and Ms. Magazine and he runs a Facebook page (Darwin’s Bulldog) that shares evolution news.
In a recent paper in the Journal of Heredity authors Liegeois, Sartori, and Schwander (2021) have proposed that mayflies (order Ephemeroptera) could provide an invaluable system for studying a long-standing question in evolutionary biology: Why sex?

It may seem an odd question to some, but the origin and maintenance of sexual reproduction has interested thinkers from Aristotle to Darwin and up to the present. At heart is the idea that sex is costly – metabolically in terms of finding mates and expending energy and genetically in transmitting half of a genome instead of the entire genome through asexual reproduction. In his “Essays on Heredity and Kindred Biological Problems”, August Weismann suggested that sexual reproduction was advantageous because it provides the variation for natural selection to act upon and allows organisms to “hedge their bets” against a changing environment. This is the explanation often taught in high school and college classrooms and is true in some cases, but it seems that more often it’s a disadvantage for successful parents to produce offspring that differ from them. This has led to a wide range of more complex genetic explanations, including Muller’s Ratchet.
The issue includes both how sexual reproduction arises and how it is maintained. A fundamental question is how a sexually reproducing individual arises and persists in an asexual population and why an asexual mutant doesn’t invade and take over a sexual population. The prevalence of sexual reproduction in nature, though, suggests that this strategy is extremely successful.
In animals, parthenogenesis (females giving rise to female offspring with no male contribution) is a not unknown asexual reproductive strategy, but one that does not seem to last long in evolutionary time. This strategy is widespread among the mayfly order, though, and Liegeois et al. suggest that this system could serve as a model system to study the costs and benefits of sexual reproduction for animals much as mixed mating systems have for plants(see Vogler & Kalisz, 2001). Mayflies are a basal insect group, and have short adult life spans and low dispersal, limiting their reproductive opportunities and possibly promoting the origin of parthenogenesis. As such they have a three distinct forms of parthenogenesis, with some species characterized by mixed mating systems.
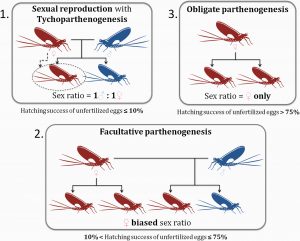
Parthenogenesis in mayflies may be accidental, facultative, or obligate. A few rare cases of deuterotokous species (where both males and females reproduce parthenogenetically) were also suggested by the literature. In obligate parthenogenesis, eggs are unable to be fertilized whereas for facultative parthenogenesis eggs may either be fertilized or unfertilized. In the interesting case when parthenogenesis is accidental (tychoparthenogenesis) the majority of the population reproduces sexually with a small percentage of eggs (less than 10%) producing offspring even when unfertilized. Importantly, species of mayflies may have a combination of reproductive strategies.
The authors surveyed the literature on mayfly reproduction analyzing a number of factors, most notably prevalence of the various reproductive strategies, phylogenetic signal, and reproductive success. Extrapolating their dataset to all know mayfly species the rate of parthenogenesis was estimated to be about 1.8%, much higher than in vertebrates but comparable to other arthropods. Taking just the species with available information the rate rose to 47.8%. Mayflies may therefore be an extreme case of parthenogenesis in arthropods, justifying the proposal that they be used as a model system for the origin and maintenance of sexual reproduction. Further, there was no evidence of phylogenetic clustering, providing a large number of phylogenetically independent contrasts to use for rigorously studying the evolution and adaptation of sex. The authors also found that fertilized eggs had a significantly higher hatching rate, explaining the fact that sexual reproduction persists despite the fact that parthenogenesis may arise regularly in this group.
“Extremely Widespread Parthenogenesis and a Trade-Off Between Alternative Forms of Reproduction in Mayflies (Ephemeroptera)” makes the argument that the mayfly order has all the components to be a fruitful model for ongoing studies in the origin and maintenance of sexual reproduction, and marshalls the evidence to support that claim. It serves as a launching pad for further work. There are several important outstanding questions that could be addressed. Researchers could take advantage of the phylogenetic contrasts to determine the environmental conditions that favor parthenogenesis and test existing hypotheses. The origin of parthenogenesis can be tracked using the range of parthenogenic strategies and mixed-mating populations. Finally, mayflies may be able to help answer why parthenogenesis is so rare and short lived despite proposed genetic advantages to this form of reproduction.
REFERENCES:



