
About the Blog Author: Nicole Conner is a Researcher III at the University of Alabama at Birmingham. Her thesis work at UAB was focused on developing both eDNA and UAV (i.e., drone) methodologies to enhance the detection of diamondback terrapins off the coast of Alabama. Her current research in Dr. Stephen Watts’ lab is focused on refining techniques for the micro-culture of the sea urchin Lytechinus variegatus for downstream applications in studies involving CRISPR-modified urchins.
While perhaps an unfamiliar concept to us as humans, multiple paternity is common in a wide range of taxa. This is when more than one male contributes genetically to a single reproductive bout of a single female. We see multiple paternity in sea turtles, fish, salamanders, and even plants. And it’s actually so widespread in snakes that it is thought to be the ancestral reproductive system (Gangloff et al. 2021).
So, what determines the occurrence of multiple paternity in a given population? How do life-history traits, reproductive strategies, ecology, and evolution interact to impact the outcomes of multiple paternity? And are there benefits or consequences to individuals and populations that have high rates of multiple paternity?
To begin answering these questions, Gangloff et al. (2021) looked to a well-established study system containing populations of western terrestrial garter snakes, Thamnophis elegans, in California, USA (Figures 1 & 2). There are two main types of habitat found around Eagle Lake in Lassen County, California and because of this, T. elegans populations in the area have diverged into two life-history ecotypes: L-fast and M-slow.
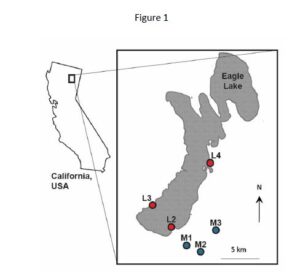
L-fast garter snake populations inhabit lakeshore habitats where food availability is reliable and predation occurs at high rates; meanwhile, M-slow snakes are found in mountain meadows where food and water availability is variable and dependent upon snow melts. As a result, the L-fast populations developed a faster pace-of-life than the M-slow populations, with faster growth rates, larger adult sizes, earlier sexual maturity, more frequent reproduction, and larger litter sizes (Gangloff et al. 2021). If there are differences in the occurrence of multiple paternity, the average number of fathers per litter, reproductive success for each sex, or reproductive skew between these two ecotypes, the researchers hypothesized that they may be able to start teasing out the details of what contributes to the patterns of multiple paternity observed in nature.

From 2006-2008, researchers collected tissue samples from adult T. elegans from three L-fast and three M-slow populations for genotyping. Snakes were sexed, measured, and weighed and gravid females were taken to Iowa State University where their litters could be born in captivity and neonates could be measured and genotyped. Purified DNA was genotyped and analyzed, with null allele frequencies and the Wahlund Effect being taken into account. A paternity analysis was conducted in order to assess differences in levels of multiple paternity between ecotypes. Finally, researchers measured reproductive success (total litter size for females and number of offspring sired for males) and made multiple estimates of reproductive skew within populations and ecotypes.
In their analysis, Gangloff et al. (2021) detected multiple paternity in all six populations. Surprisingly, females from both L-fast and M-slow ecotypes had an average of 1.5 sires contributing to each litter and the number of multiply-sired litters didn’t differ between them or among individual populations. Additional analyses showed that neither maternal size nor litter size seemed to affect the occurrence of multiple paternity. This is all despite the fact that reproductive success in both sexes was higher in the L-fast ecotype and reproductive skew as well as variance in reproductive success were greater in M-slow males than L-fast males.
These results allowed Gangloff et al. (2021) to rule out several contributing factors for multiple paternity in T. elegans populations, such as female size and female reproductive output. Instead, they suggest that males and females are employing different reproductive strategies between the ecotypes. For example, M-slow females may be employing pre- and post-copulatory mechanisms to compensate for their smaller litter sizes. These could include bet-hedging (especially considering they live in more stochastic environments than L-fast females) and mating more often with less choice than L-fast females. They also propose that there is strong intrasexual competition among males, especially in the L-fast ecotype.
Like many studies of this nature, the researchers seem to leave with more questions than straightforward answers; however, they have narrowed the wide field of possible explanations and can now direct their attention in specific areas. Gangloff et al. (2021) pose that studies investigating sperm competition, female reproductive strategies, behavioral differences among males, and potential benefits incurred by females from multiple paternity could further reveal the web of interactions that lead to the observed patterns of multiple paternity in these T. elegans populations.
References



