
About the blog author: Allison K. Miller is a conservation biologist and PhD candidate in the Gemmell Lab at the University of Otago doing research on lamprey genetics in the Southern Hemisphere. She studied coral reef ecology at the University of Guam where her master’s thesis research focused on holothuroid (sea cucumber) phylogenetics. She then worked as a biological technician for the US National Park Service in Guam. You can follow her on Twitter @AMillerH20bear7 or via her lab’s website.
In the rivers of the cool fern forests of Aotearoa New Zealand lives a mysterious fish. It is nocturnal and moves most on rainy nights. It is a determined species that will go to extreme limits to complete its migration upstream — at times it will even climb out of water!
This elusive creature is the pouched lamprey (Geotria australis), or kanakana/piharau as some of the names they are known as in te reo Māori (the Māori language). Kanakana/piharau is a culturally and ecologically important lamprey species and is the most widespread of the five lamprey species found in the seas, rivers, and lakes of the Southern Hemisphere (Riva-Rossi et al. 2020; Miller et al. 2021). Kanakana/piharau have an anadromous life cycle that is similar to salmon, in that they breed in fresh water but spend much of their lives in the ocean. Larvae hatch in fresh water rivers and streams and will burrow and filter feed there for approximately four years until they metamorphose into juveniles. The juveniles will then migrate downstream to the ocean where they will spend a certain amount of time feeding — we still do not know for certain how long or what they prey on — until they are full grown. These full grown adults (up to 750 mm) will then stop feeding and begin the long perilous migration back into freshwater rivers. They will migrate in vast numbers, often hundreds or even thousands at a time! Once upstream, male and female pairs build nests and guard their ~55,000 eggs until they hatch (Baker et al. 2017). These ultimate parents will then die after ensuring the safe hatching of their offspring.
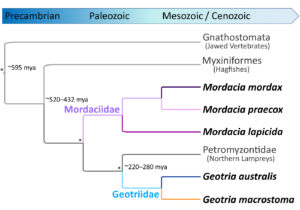
Southern lamprey, like their ~36 northern relatives, are venerable survivors of an ancient lineage that dates back close to the dawn of Vertebrata, perhaps originating more than 360 million years ago (Docker et al. 2019; Miyashita et al. 2021; Figure 1).
However, despite being a long surviving lineage, lamprey are often negatively perceived. This is mostly because one species, Petromyzon marinus, was accidentally introduced into the Great Lakes where it became an abundant and well-known pest (Great Lakes Fishery Commission 2000). In addition, these jawless fishes have a unique way of feeding — they will latch onto their prey in a so called “sucking” fashion. This has produced some rather unbecoming photos of lamprey who have been caught with their oral-disk (mouths) wide open.
Such as this as this poor fella (Figure 2a).

Ask any lamprey and they will probably agree that this is not their most attractive angle.
Despite their fearsome “teeth”, kanakana/piharau pose no danger to humans and in fact, they do not feed during their ~15 month freshwater breeding migration (Miller et al. 2021). They also have a beautiful blue colouration when they are juveniles (see Figure 3).
Kanakana/piharau are considered a taonga, or treasured species, to many iwi (Māori tribes) and iwi in New Zealand have collected them for hundreds of years, developing elaborate fishing mechanisms to do so (Figure 4). Kanakana/piharau are also the focus of Māori proverbs that boast of their determination and strength. Ecologically, kanakana/piharau in New Zealand are important prey for many predators, including endangered species such as the yellow-eyed penguin/hoiho, one of the rarest penguins in the world. For those not captured during their upriver migration, adult lamprey die after spawning and add important marine nutrients into the freshwater environment.
In 2012 there were reports of Lamprey Reddening Syndrome causing mass lamprey mortalities in the South Island of New Zealand. Lamprey Reddening Syndrome — of which the cause is still unknown — causes haemorrhaging, lesions, and boils on the bodies of kanakana/piharau and is often fatal. This had kaitiaki (iwi caregivers/guardians) and managers all over New Zealand concerned as kanakana/piharau in New Zealand were, and still are, considered a “Threatened — nationally vulnerable” and declining species. Lamprey Reddening Syndrome was only reported from the South Island of New Zealand, but managers and kaitiaki feared that it would spread to the North Island. However, it was difficult to incorporate protection measures for Lamprey Reddening Syndrome into management plans because very little was known about the migrations and population structure of kanakana/piharau in New Zealand. In particular, managers/kaitiaki wished to know whether there was one large inter-breeding population? Or, were there multiple smaller populations found in certain areas of the country that warranted management as separate entities?

This is where we came in. Despite kanakana/piharau being elusive, our talented iwi collaborators and scientists from the National Institute of Water and Atmospheric Research (NIWA) worked together to sample over 300 lamprey across 10 locations in New Zealand — the largest collection ever for this species! We then analysed both multi-locus genetic data (RADSeq) and targeted gene (COI and Cyt-b) data from these samples to acquire the population structure information needed by managers/kaitiaki to inform lamprey reddening syndrome management plans.

Reduced-representation sequencing, or RADSeq, is a technique that uses restriction enzymes to digest genomic DNA into certain size fragments which are then sequenced and used to find genotypes and single nucleotide polymorphisms (SNPs). The SNPs from individuals from different populations can then be compared and used to identify their relationships, or population structure. Unlike targeted gene sequencing, RADSeq can obtain information from the entire genome but without the cost of having to sequence the entire genome of the organism. However, we did also sequence the entire genome of one kanakana/piharau to identify outlier SNPs and assist future management efforts. Using both the multi-gene and targeted-gene techniques we found that kanakana/piharau from New Zealand were all one population (Figure 5).
This finding is important because it tells us that kanakana/piharau can likely be managed as one population, which has implications for possible management efforts such as translocations and propagation facilities. Unfortunately, the findings also suggest that if Lamprey Reddening Syndrome is transmittable to other individuals, it has a high chance of affecting kanakana/piharau on the North Island. However, with this information managers/kaitiaki can now be aware of the potential of Lamprey Reddening Syndrome to spread to the North Island and they can implement monitoring methods for early detection.
This was a joint effort that would not have been possible without the help of our iwi collaborators. Iwi hold invaluable knowledge, or mātauranga Māori, about the genealogy, or whakapapa, of kanakana/piharau and their contributions greatly assisted this study. We are grateful for their help and the lamprey that made this study possible. We anticipate this information to now be incorporated by managers and kaitiaki into conservation strategies to support the well-being of this mysterious and treasured species.
Additional resources available at:
Restoration and enhancement of piharau/kanakana/lamprey
Murihiku kanakana/lamprey monitoring
References:
Kuraku S, Ota KG, Kuratani S (2009) Jawless fishes (Cyclostomata). In Hedges SB, Kumar S (eds) The timetree of life. Oxford University Press, Oxford. pp. 317–319.



