
About the author: Nisha Dwivedi a conservation biology masters student at Lund University. As part of her thesis project she is currently exploring genetic variation at the immune gene level in different rodent species.
When thinking of pathogens, we usually imagine bacteria, viruses, or even parasites. Indeed, despite their important differences, most pathogens contain some form of nucleic acid. An exception to this pattern comes in the form of prions. It is in fact their form itself that makes them infectious. As you probably already know, from a simple chain of amino acids, proteins can take on more and more complex structures. The final structure of a protein is vital to its action and changes to this structure can transform normal and functional proteins into prions. In other words, prions are pathogenic agents with the same amino acid sequences as proteins, but with a different conformation.
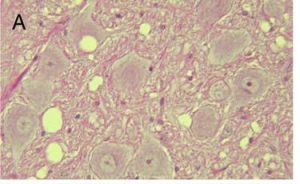
When a prion enters a host, it interacts with normal prion proteins to convert these into infectious prions. These prions then accumulate in the host’s central nervous system. In fact, by looking at histological samples of an infected individual’s nervous tissue, you can actually see these masses of prions (see Figure 1). This leads to degenerative brain disorders, also known as transmissible spongiform encephalopathies (TSE). Today, we know about several different TSEs that impact very different groups of organisms. Mad-cow disease comes to mind.
Chronic wasting disease (CWD) is a TSE affecting members of the cervid family. The disease was first recognised in the 1960s amongst mule deer in a Colorado Research Facility. It was only in 1978 that prions were identified as the pathogenic agents responsible. The specific gene encoding the normal protein is known as the prion protein gene (PRNP). With increasing research, it became evident that the disease can affect a wide range of deer species and that CWD prions are quite readily exchanged between different species. It was also realized that the infections were not exclusive to captive populations, as CWD was also detected in wild populations. Bodily fluids, such as saliva, semen, urine, and blood, are what allow prions to be transmitted between individuals. When an animal becomes infected with CWD prions, the disease will progressively develop and cause weight loss, excessive salivation, but also behavioural changes such as a loss of fear of humans (Belay et al., 2004; Perrin-Stowe et al., 2020; Telling, 2013; Sakudo, 2020).

Given there is no cure for TSEs and management of CWD hasn’t always proved successful, deer species are facing great risks. The Florida Key deer (Odocoileus virginianus clavium) and the Columbian white-tailed deer (Odocoileus virginianus leucurus) are two sub-species of white-tailed deer currently classified as endangered in North America. Both have faced overhunting in the past and, though CWD has not yet reached these populations, the consequences if it did could be devastating. In a study of 2433 mainland white tailed deer, variations in the PRNP gene were shown to be associated with different propensities to CWD. Several haplotypes of the gene were identified and some were shown to be significantly less frequent in infected individuals in comparison to non-infected individuals (Brandt et al., 2018). In this context, a recent study focused on identifying haplotypes in the two endangered white-tailed deer subspecies to estimate their susceptibilities to CWD (Perrin-Stowe et al., 2020).
In their study, Perrin-Stowe et al. (2020) analysed a total of 61 samples. Forty-eight of these samples were of Florida Key deer and the other 13 were of Columbian white-tailed deer. By sequencing the PRNP gene, the researchers found three haplotypes of this gene in the Florida Key deer and two in the Columbian white-tailed deer. The three Florida Key deer haplotypes differed by 5 polymorphisms. Of the three haplotypes, the two most frequent share a non-synonymous mutation at codon 96. This same mutation was associated to lower CWD frequencies in the large-scale study of mainland white-tailed deer. The two Columbian white-tailed deer haplotypes only differed by one synonymous mutation. The identical amino acid sequences given by these two haplotypes were also found in high frequencies within the infected mainland white-tailed deer individuals.

These results show that haplotypes associated with reduced vulnerability to CWD are quite common in the Florida Key deer. The Columbian white-tailed deer, on the other hand, showed no evidence of haplotypes associated with lower vulnerabilities to CWD. If CWD were to reach both populations, the Florida Key deer might therefore show a greater resistance than the Columbian white-tailed deer. Given the current spread of CWD across the continent, particular attention should be paid to Columbian white-tailed deer in management efforts. The authors also suggest that selective breeding of less vulnerable Florida Key deer individuals could further protect an already vulnerable population (Perrin-Stowe et al., 2020).
References



